JRI Ingeniería Optimizes Document Management and Project Traceability with Accruent Meridian
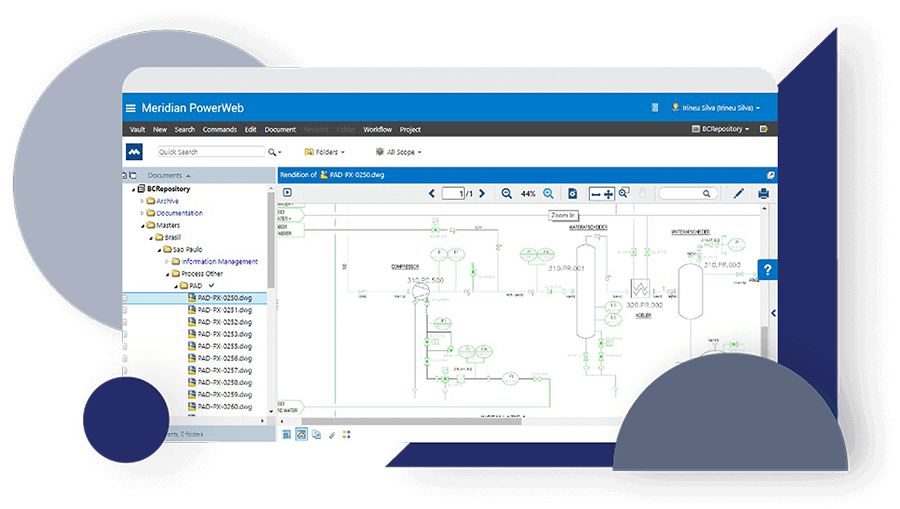
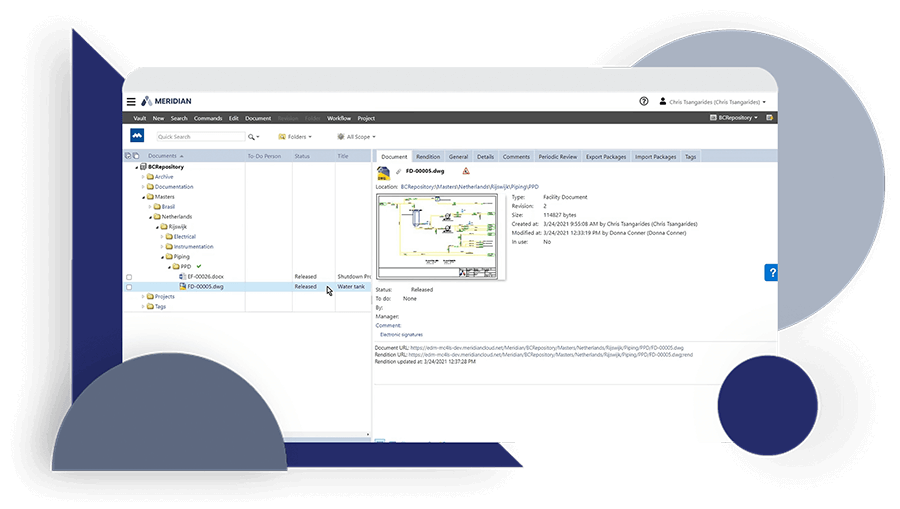
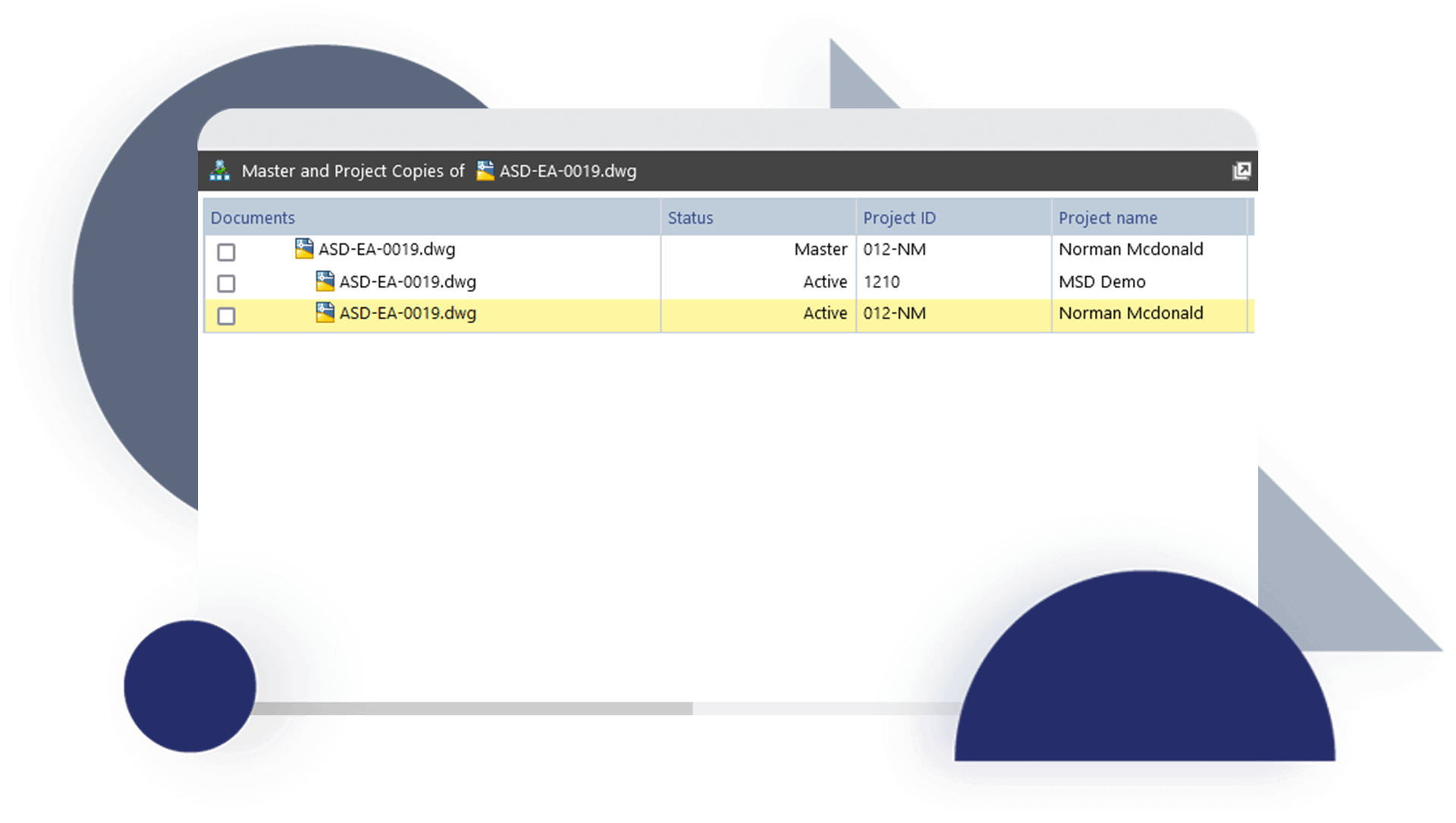
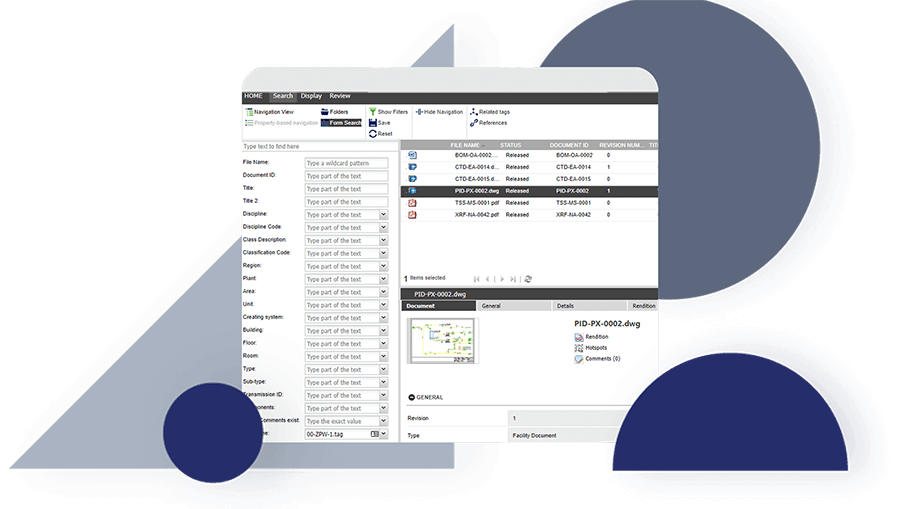
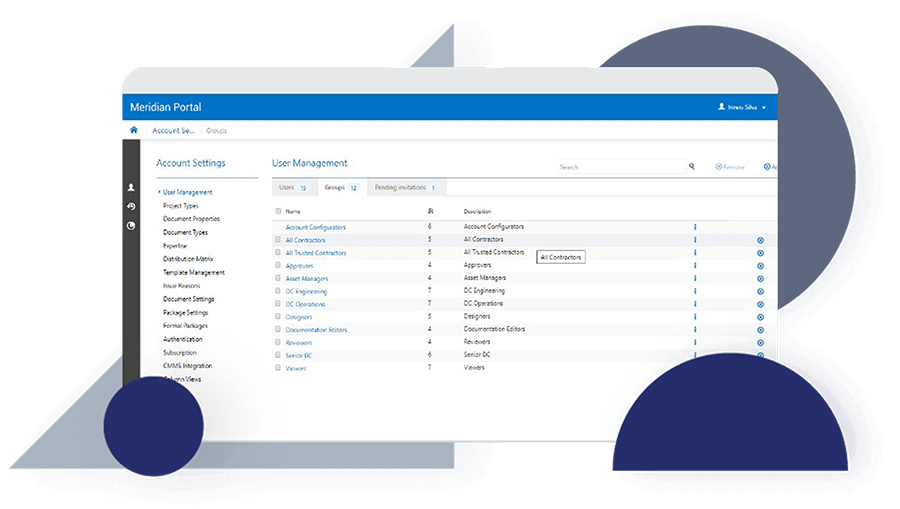
Discover how JRI Ingeniería improved the traceability of its deliverables, reduced errors caused by duplication and increased productivity in its mining engineering projects with Accruent Meridian, together with the support of Microgeo Ingeniería.



.png)







